
Welcome to the Bhargava Laboratory
Research
Understanding the pathophysiology of Multiple Sclerosis to identify new treatment strategies

Metabolomics in MS
There is a major unmet need for biomarkers of MS disease progression. Our lab is currently focusing on the utility of new “omics” technologies such as metabolomics and lipidomics in identifying both prognostic and diagnostic biomarkers for MS. Metabolomics and lipidomics refer to the use of mass spectrometry coupled with liquid or gas chromatography to identify levels of multiple metabolites / lipid species in biological matrices such as plasma and CSF. Current projects involve utilizing metabolomics to identify a metabolic signature of MS, identifying metabolic pathways that are related to progression in MS and utilizing metabolomics to understand the mechanism of action of the disease modifying agent dimethyl fumarate. We are also tracking a large cohort of patients longitudinally to determine whether serum lipidomic profiles predict progression of disease over time. These studies could help better understand the pathophysiology of the disease and provide markers for use in the testing of new therapies for progressive MS.
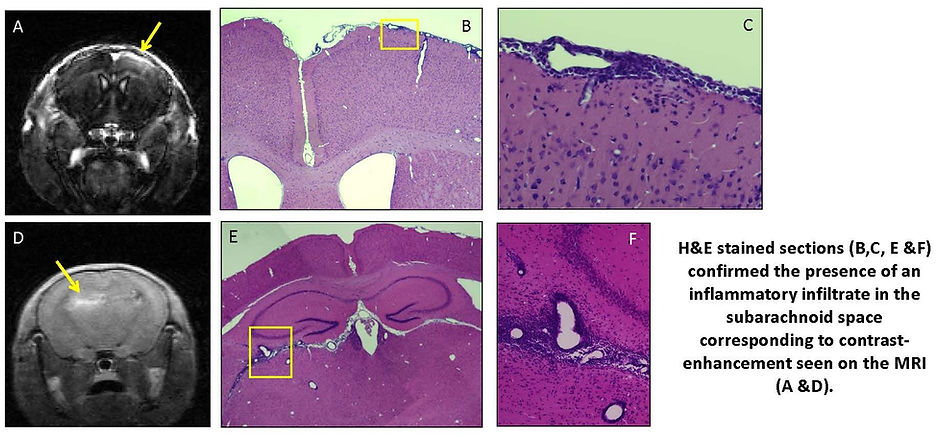
Leptomeningeal inflammation in MS
Leptomeningeal inflammation has been noted in relapsing remitting MS as well as progressive MS and has been associated with a more severe disease course and increased cortical demyelination. B cells are an important component of these meningeal infiltrates and in a subset of MS patients, ectopic lymphoid follicle like structures can be identified in the meninges. Meningeal inflammatory infiltrates in MS patients can now be detected on MRI imaging providing a possible marker of therapeutic response for trials targeting this process. However, the testing of potential therapeutic agents is hampered by the lack of a suitable animal model.
We performed ultra-high field MRI (11.7 T) in mice with relapsing-remitting EAE late in their disease course and identified meningeal contrast enhancement that pathologically corresponds to meningeal inflammation. These meningeal infiltrates are composed primarily of B and T cells and are associated with demyelination and axonal damage in the adjacent cortex. These lesions also persist and accumulate over time providing a potential marker to test the efficacy of therapies targeting meningeal inflammation. We are currently in the process of further defining the effects of meningeal inflammation on the adjacent cortex and testing new therapeutic agents that could impact the various components of the meningeal infiltrates.

CNS cell-derived Extracellular vesicles in MS
Extracellular vesicles (EVs) are produced by all cells in the body and play important physiological and pathological roles in intercellular communication. Since EVs express proteins on their surface derived from their cell of origin, multiple groups have developed strategies to isolate EV subsets enriched for a particular cell of origin. For example, L1-CAM is an adhesion molecule primarily expressed on neurons and L1-CAM+ EVs are highly enriched in neuronal proteins suggesting that this sub-set of EVs is enriched for neuronal origin. Similarly, GLAST has been identified as a potential marker to identify astrocyte-derived EVs. We partnered with colleagues at the National Institutes of Aging to apply this method to people with MS to help identify a new platform for biomarker discovery and understanding MS pathophysiology.
We noted in a study of 61 people with MS and 31 healthy individuals that neuronally-enriched EVs (NEVs) from people with MS has markedly reduced levels of synaptic proteins and AEVs from people with MS had elevated levels of multiple complement components compared to healthy individuals. These were interesting findings since pathological studies have demonstrated significant synaptic loss in MS and recent studies have suggested that synaptic loss is mediated by complement deposition on these synapses leading to removal by microglia. Most remarkably, we also showed in this study that there was as striking inverse correlation between levels of NEV synaptic proteins and a number of AEV complement component levels suggesting that higher astrocyte complement production drives synaptic loss in MS.
We are currently funded by the Myelin Repair Foundation to evaluate longitudinal changes in these NEV and AEV markers in people with MS and to link them with imaging markers of neurodegeneration. We are also funded to assess longitudinal changes in a broad range of biomarkers in EV subsets in a large cohort of controls, people with RRMS and PMS. We plan to use these EV subset-based biomarkers to monitor effects of therapeutic interventions - such as the effect of TUDCA on AEV complement in our ongoing trial of TUDCA in PMS.
Additionally, we are in the process of identifying markers of OPC- and oligodendroglia-derived EVs to interrogate ongoing damage or repair within these cell populations, especially in trials of putative remyelinating agents.

Selected Publications
The Fruits of Our Labor
May 2021
Imaging meningeal inflammation in CNS autoimmunity identifies a therapeutic role for BTK inhibition
https://academic.oup.com/brain/article/144/5/1396/6173632
June 17, 2020
Synaptic and complement markers in extracellular vesicles in multiple sclerosis
https://journals.sagepub.com/doi/full/10.1177/1352458520924590
March 2020
Bile acid metabolism is altered in multiple sclerosis and supplementation ameliorates neuroinflammation
https://www.jci.org/articles/view/129401
November 2021
Discordant humoral and T cell immune responses to SARS-CoV-2 vaccination in people with multiple sclerosis on anti-CD20 therapy

Lab Members
A Collaborative & Diverse Group

Lab Alumni
Ashley Muller (Clinical Research Coordinator)
Dimitrios Ladakis (Clinical Post-Doctoral Fellow)
Kanak Gupta (Research Technologist)
Steven Rua (Undergraduate Researcher)
Sophia Kim (Research Technologist)
Sakshi Chopra (Clinical Research Coordinator)
Ajay Kesharwani (Research Post-Doctoral Fellow)
News
Latest developments in the Bhargava Lab
Our work highlighted by CDMRP
Kanak accepted to Graduate school
Kanak will begin her program at University of Toronto in the Fall. Congratulations !! We will miss you.
Job Openings
Research Opportunities at Bhargava Laboratory
October 12, 2020
Research Coordinator/ Research Fellows
We are looking for candidates interested in working in our research group on the clinical side. We are currently beginning two investigator-initiated trials and you would be involved in getting these off the ground and recruiting/ following patients in these trials. Recent grads/ IMG/ FMGs are welcome to apply. Please contact me at pbharga2@jhmi.edu.
October 15, 2020
Postdoctoral Fellow
We are looking for candidates interested in working in the laboratory on new porojects related to leptomeningeal inflammation in MS or EVs in MS. Prior experience with animal models of disease and in vitro work with neuronal/ glial cells would be a plus. Please contact me at pbharga2@jhmi.edu.
Contact Us






_edited.jpg)

